Key Takeaways
When selecting polymers for surgical instruments, understanding critical material properties and regulatory requirements ensures optimal performance and patient safety.
- Sterilization resistance is paramount – Polymers must withstand repeated autoclave cycles at 121-134°C without losing mechanical properties or dimensional stability
- PEEK dominates high-performance applications – Offers 3,000+ sterilization cycles, bone-matching elasticity, and complete radiolucency for imaging compatibility
- Match polymer to specific instrument needs – Load-bearing tools require high tensile strength materials, while flexible devices need elastomeric properties
- Biocompatibility testing follows ISO 10993-1 – Contact duration and tissue exposure determine the depth of required biological safety evaluation
- Post-sterilization validation is essential – Comprehensive testing of dimensional accuracy and mechanical strength after repeated cycles prevents premature failure
The key to successful polymer selection lies in balancing sterilization durability, biocompatibility, and mechanical performance against specific surgical application requirements and regulatory compliance standards.
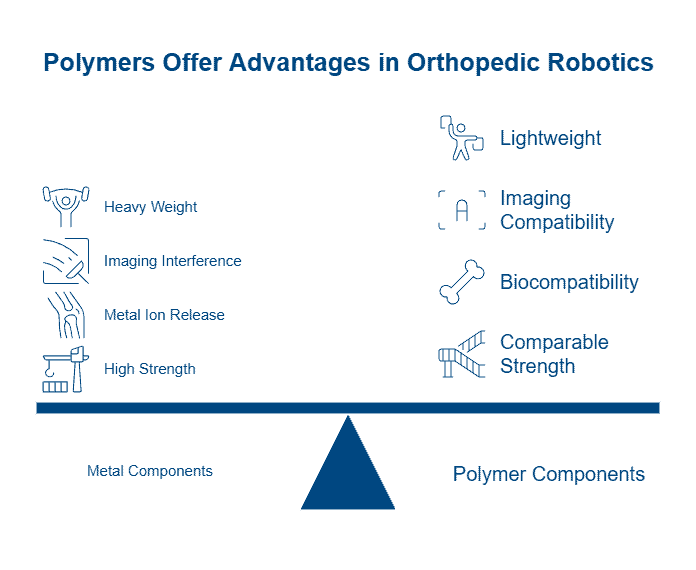
Polymers offer a compelling advantage for surgical instruments, weighing up to ten times less than typical metals while delivering comparable strength and durability. This weight reduction, coupled with superior chemical resistance, has driven widespread adoption of polymers for surgical instruments across the medical device industry. However, selecting the right material requires careful evaluation of multiple factors.
Medical grade polymers must meet stringent biocompatibility standards, withstand repeated sterilization cycles, and maintain dimensional stability under demanding conditions. Properties such as chemical resistance, radiolucency, and resistance to hydrolysis can determine whether a polymer succeeds or fails in surgical applications. This guide examines the critical material properties that matter when choosing high-performance polymers for medical devices and provides practical insights for selecting biocompatible polymers that meet regulatory requirements.
Critical Material Properties for Surgical Instrument Polymers
Sterilization resistance stands as the most demanding requirement for polymers used in medical devices. Steam autoclaving, which exposes materials to saturated steam at 121°C for 30 minutes or 132°C for 3-4 minutes, remains the most widely used sterilization method. High-performance polymers must withstand these extreme conditions repeatedly without significant loss of mechanical properties or dimensional changes.
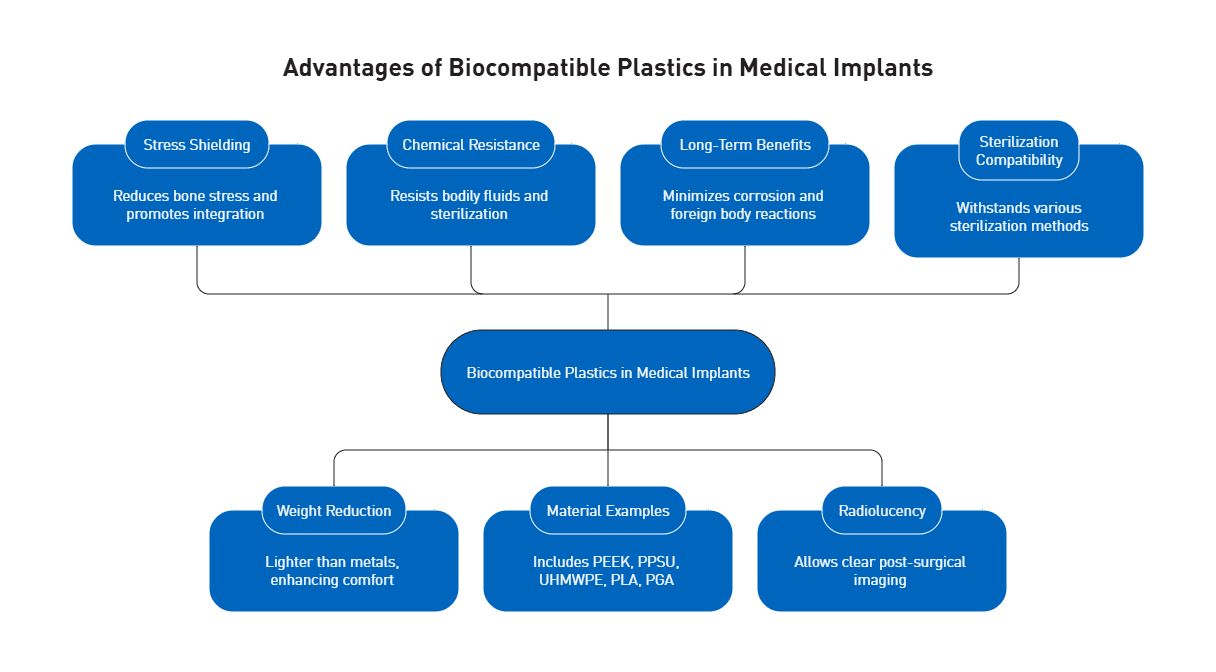
Biocompatibility represents another non-negotiable requirement. Medical grade polymers must meet ISO 10993-1 standards, which provide a risk-based framework for evaluating biological safety based on contact duration and device type. This standard has replaced older USP Class VI requirements, offering a more rigorous evaluation system that identifies and quantifies chemical constituents in materials.
Chemical resistance determines long-term durability. Surgical instruments face frequent exposure to aggressive disinfectants including quaternary ammonium compounds, sodium hypochlorite, hydrogen peroxide, and phenolic solutions. Polymers lacking adequate chemical resistance develop environmental stress cracking, surface defects, and premature failure.
Hydrolysis resistance protects against degradation from hot steam vapor. Materials sensitive to hydrolysis can experience polymer chain scission, leading to brittleness and structural deterioration after repeated autoclave cycles. Correspondingly, dimensional stability ensures instruments maintain precise tolerances through hundreds of sterilization cycles, which proves critical for surgical accuracy and device performance.
Radiolucency provides transparency to x-rays for specific applications, enabling surgeons to visualize anatomy without removing instruments during fluoroscopic procedures.
Top High-Performance Polymers for Surgical Instruments
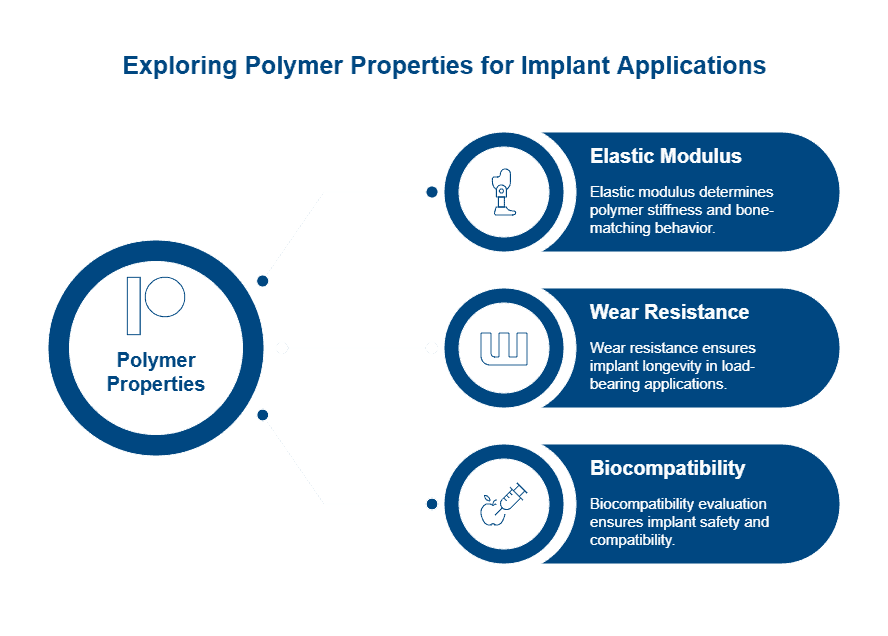
PEEK (polyether ether ketone) dominates medical grade polymers for demanding surgical applications, withstanding continuous temperatures up to 260°C and maintaining properties through more than 3,000 high-pressure sterilization cycles at 134°C. The material’s melting point reaches 343°C, while its elastic modulus closely matches human bone, reducing stress shielding in load-bearing applications. Notably, PEEK renders completely transparent on X-rays, CT, and MRI scans, enabling precise surgical guidance without imaging interference.
Polyphenylene sulfide (PPS) provides a cost-effective alternative to PEEK for moderate-temperature applications. This semi-crystalline thermoplastic maintains chemical resistance and tensile strength even above 200°C, with continuous service capability reaching 230°C depending on load. Glass fiber reinforced grades, such as PPS GF40 with 40% glass fiber content, enhance mechanical strength for surgical instrument components.
Radel PPSU (polyphenylsulfone) excels in sterilization durability, surviving over 1,000 autoclave cycles at 134°C while maintaining dimensional stability. Medical device manufacturers achieve up to 50% weight reduction compared to metal instruments using Radel PPSU, which meets ISO 10993 biocompatibility standards for limited bodily fluid contact.
Ultem PEI delivers exceptional mechanical strength among amorphous thermoplastics, with a glass transition temperature of 217°C. The polymer withstands hundreds of sterilization cycles through steam, gamma radiation, and ethylene oxide, making it suitable for reusable surgical instruments and autoclavable trays.
Selecting Medical Grade Polymers Based on Instrument Requirements
Application requirements dictate polymer selection more than any single material property. Load-bearing surgical instruments demand polymers with high tensile strength and stiffness, while flexible devices require elastomeric properties. Contact duration with bodily tissues determines the depth of biocompatibility testing required under ISO 10993 standards.
Sterilization method compatibility shapes material choices for reusable instruments. PPSU withstands over 1,000 steam autoclave cycles without significant property loss, making it ideal for instruments requiring extended service life. Conversely, single-use instruments benefit from glass-reinforced PARA compounds optimized for gamma radiation sterilization, which show no significant appearance or performance changes after exposure.
Regulatory compliance requirements vary by market and device classification. Medical device manufacturers must adhere to Quality System Regulation 21 CFR Part 820 and demonstrate ISO 13485 certification for market entry. Biocompatibility testing follows ISO 10993-1 protocols, evaluating physical and chemical characteristics plus exposure parameters including contact nature, degree, frequency, and duration.
Material validation protocols require comprehensive post-sterilization testing. Manufacturers must evaluate dimensional accuracy, mechanical strength, and surface finish after repeated sterilization cycles. Some composites experience 17% to 57% decreases in short-beam strength after 400 sterilization cycles, consequently requiring careful assessment of performance thresholds to optimize surgical workflows.
Conclusion
Choosing the best polymers for surgical instruments is ultimately a matter of matching material performance to clinical function, sterilization demands, and regulatory expectations. High-performance materials such as PEEK, PPS, PPSU, and PEI each offer valuable advantages, but the right choice depends on how the instrument will be used, how often it will be sterilized, what chemicals it will encounter, and what mechanical tolerances it must maintain over time.
For medical manufacturers, the strongest results come from a material selection process that considers the full application environment, not just isolated datasheet values. When supported by post-sterilization validation and precision manufacturing expertise, the right polymer can improve instrument reliability, extend service life, and help meet the performance standards today’s surgical applications demand.
Selecting the right material is only part of the equation. Precision machining, contamination control, and application-specific engineering support are just as critical when developing surgical instruments for real-world medical use.
AIP Precision Machining works with medical device manufacturers to machine high-performance polymers for demanding applications that require tight tolerances, repeatable quality, and regulatory-minded production. If you are evaluating polymers for surgical instruments, contact AIP to discuss your design, material requirements, and manufacturing goals.
FAQs
Q1. What are the most important properties to consider when selecting polymers for surgical instruments?
The most critical properties include sterilization resistance (particularly to steam autoclaving at 121-132°C), biocompatibility meeting ISO 10993-1 standards, chemical resistance to disinfectants, dimensional stability through repeated sterilization cycles, and mechanical strength suitable for the instrument’s intended use. For certain applications, radiolucency for imaging compatibility is also essential.
Q2. Which polymer is best for load-bearing surgical instruments?
PEEK (polyether ether ketone) is the top choice for load-bearing surgical applications. It can withstand continuous temperatures up to 260°C, maintains properties through more than 3,000 sterilization cycles at 134°C, and has an elastic modulus that closely matches human bone. Additionally, PEEK is completely transparent on X-rays, CT, and MRI scans.
Q3. How many sterilization cycles can high-performance medical polymers withstand?
The durability varies by polymer type. Radel PPSU can survive over 1,000 autoclave cycles at 134°C while maintaining dimensional stability. PEEK performs even better, withstanding more than 3,000 high-pressure sterilization cycles at 134°C. Ultem PEI can endure hundreds of sterilization cycles through various methods including steam, gamma radiation, and ethylene oxide.
Q4. What biocompatibility standards must medical grade polymers meet?
Medical grade polymers must meet ISO 10993-1 standards, which provide a risk-based framework for evaluating biological safety based on contact duration and device type. This standard has replaced older USP Class VI requirements and offers a more rigorous evaluation system that identifies and quantifies chemical constituents in materials.
Q5. Why is chemical resistance important for surgical instrument polymers?
Chemical resistance is crucial because surgical instruments face frequent exposure to aggressive disinfectants including quaternary ammonium compounds, sodium hypochlorite, hydrogen peroxide, and phenolic solutions. Polymers lacking adequate chemical resistance can develop environmental stress cracking, surface defects, and premature failure, compromising the instrument’s long-term durability and safety.





 Fuel system components
Fuel system components




 The selection of materials is crucial in the manufacturing of orthopedic implants, as these materials must offer exceptional biocompatibility, strength, and durability to withstand the rigors of the human body.
The selection of materials is crucial in the manufacturing of orthopedic implants, as these materials must offer exceptional biocompatibility, strength, and durability to withstand the rigors of the human body.
 In the highly regulated field of medical device manufacturing, rigorous quality control and strict adherence to regulatory standards are essential. Ensuring that every product meets the highest levels of safety, performance, and reliability is a critical responsibility.
In the highly regulated field of medical device manufacturing, rigorous quality control and strict adherence to regulatory standards are essential. Ensuring that every product meets the highest levels of safety, performance, and reliability is a critical responsibility.